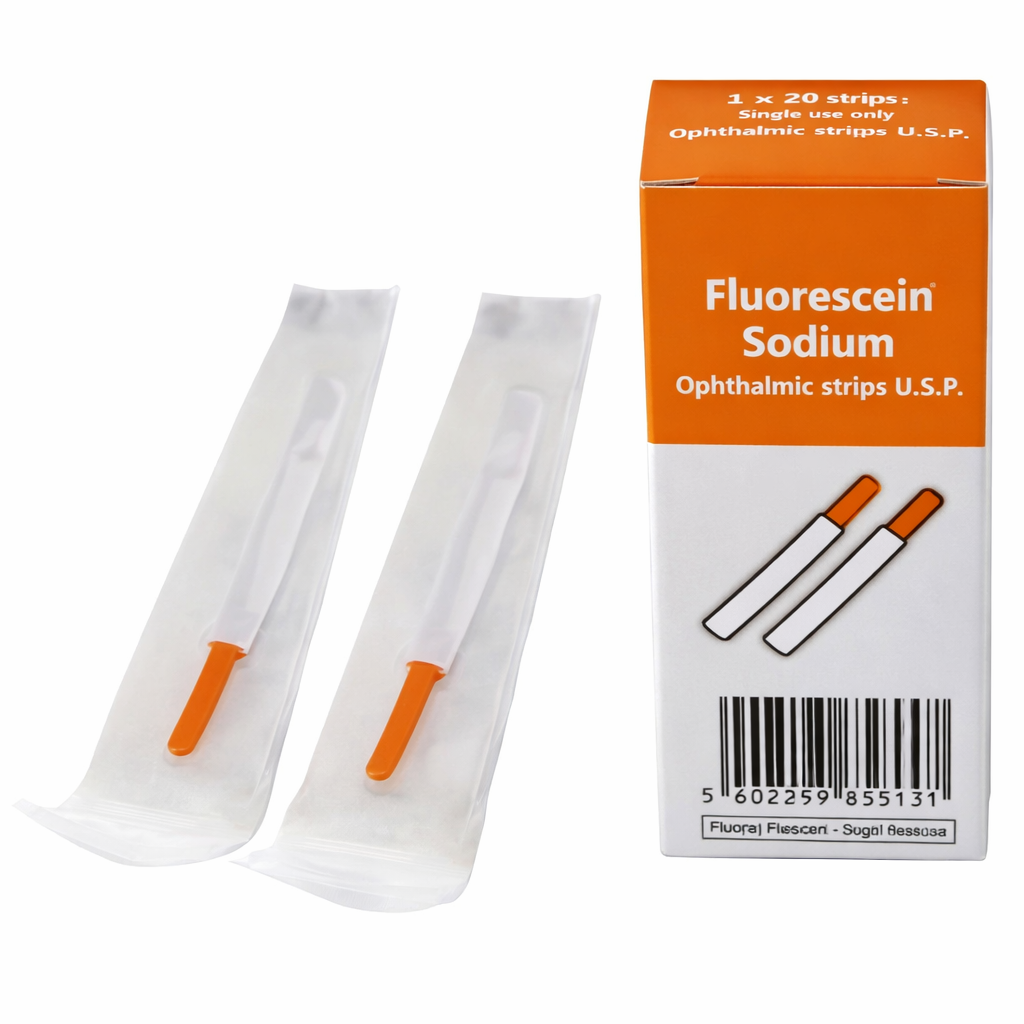
Fluorescein Sodium Strips: The Gold Standard in Ophthalmic Diagnostics—Exporting to Dubai, Muscat, Abuja, and Beyond
In the precision-driven world of ophthalmology, the quality of diagnostic tools directly impacts patient outcomes. Ernest Vision, a specialized division of Ernest Pharmaceutical Pvt. Ltd., is a leading global exporter of high-quality Fluorescein Sodium Strips.
We provide seamless supply chain solutions to eye clinics, hospitals, and distributors in Dubai (UAE), Muscat (Oman), Abuja (Nigeria), Yaoundé (Cameroon), and Jakarta (Indonesia).
What are Fluorescein Sodium Strips?
Fluorescein Sodium Strips are essential diagnostic agents used to visualize corneal integrity and fit contact lenses. When moistened with sterile water or saline and applied to the eye, the dye reveals abrasions, foreign bodies, and tear film efficiency under a blue cobalt filter.
Product Highlights & Specifications
| Feature | Details |
| Active Agent | Fluorescein Sodium (USP Grade) |
| Concentration | 1.0 mg per strip |
| Sterility | Individually wrapped sterile pouches |
| Indication | Diagnostic staining of the anterior segment |
| Box Quantity | 100 to 300 strips (Bulk options available) |
Why Source Ophthalmic Strips from Ernest Vision?
As a dedicated Ophthalmic Device Exporter, we bridge the gap between advanced Indian manufacturing and global healthcare demands.
1. Market-Specific Expertise
Dubai: Meeting the high regulatory standards of the Middle East with rapid air-freight logistics.
2. Quality Assurance
Our strips are manufactured in ISO and GMP-certified facilities, ensuring non-toxic, medical-grade paper that prevents irritation while providing high-contrast fluorescence for clear diagnosis.
3. Bulk Supply & OEM Partnerships
Whether you are a private hospital in Jakarta or a government distributor in Abuja, we offer flexible shipping and private labeling (OEM) to suit your regional market needs.
Technical Product Profile: Fluorescein Sodium Strips
Fluorescein Sodium is a vital dye belonging to the Xanthine group.
1. Key Features & Manufacturing Standards
Pharmaceutical Grade Dye: Impregnated with approximately 1.0 mg of Fluorescein Sodium USP.
Substrate: High-quality, medical-grade lint-free cellulose fiber paper.
Sterilization: Each strip is individually wrapped and sterilized using Ethylene Oxide (EO) or Gamma Irradiation to ensure zero microbial contamination.
Safety Profile: Non-toxic, non-irritating, and physiological pH-balanced to ensure patient comfort.
Certifications: Manufactured in GMP-certified facilities; complies with ISO 13485:2016 and CE standards.
2. Clinical Applications
Beyond simple staining, these strips are used for complex diagnostic procedures:
Applanation Tonometry: Used to create the "fluorescein semicircles" required to measure intraocular pressure (IOP) for glaucoma screening.
Tear Film Break-up Time (TBUT): Evaluates tear film stability and identifies Dry Eye Disease (DED).
Corneal Integrity Mapping: Highlights corneal abrasions, ulcers, herpes simplex keratitis, and foreign bodies.
Contact Lens Fitting: Essential for assessing the relationship between a rigid gas-permeable (RGP) lens and the cornea (fluorescein pattern).
Siedel Test: Detects aqueous humor leakage from surgical incisions or penetrating trauma.
Guidelines for Use
To ensure maximum fluorescence and patient safety, the following protocol is recommended:
Preparation: Check the expiry date and ensure the sterile pouch is intact.
Moistening: Moisten the impregnated tip with 1–2 drops of sterile saline or irrigation solution.
Note: Avoid touching the tip of the saline bottle to the strip. Application: Ask the patient to look up. Gently touch the moistened tip to the inferior tarsal conjunctiva (inside the lower eyelid). Avoid direct contact with the cornea to prevent accidental scratching.
Distribution: Allow the patient to blink several times to spread the dye evenly across the tear film.
Visualization: Observe the eye under a Slit Lamp using a Cobalt Blue Filter.
For enhanced contrast, use a yellow (Wratten) filter in conjunction with the blue light.
Storage & Handling for Export
Because Ernest Vision exports to diverse climates (from the humidity of Jakarta to the heat of Dubai), storage is critical:
Temperature: Store in a cool, dry place below 30°C.
Light Sensitivity: Protect from direct sunlight as the dye can degrade over time.
Moisture Control: Keep in original heat-sealed pouches; moisture can cause the dye to bleed before use.
Why Choose "Ernest Vision" as an OEM Partner?
Customization: We offer private labeling and custom packaging for distributors in Oman, Nigeria, and Cameroon.
Bulk Availability: Large-scale stock ready for immediate dispatch to Abuja and Jakarta.
Documentation: We provide full COA (Certificate of Analysis) and MSDS for smooth customs clearance in the UAE.
📞 Contact Ernest Vision – Ophthalmic Device Exporter
(GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.)
We are your trusted partner for Ophthalmic diagnostics, Oncology medications, and general pharmaceutical exports. Reach out today for a custom quote or dealership inquiry.
🌐 Explore Our Global Portals:
www.ernestvision.com www.ernestimpex.com www.oncologymedicinesupplier.com
📧 Professional Inquiry:
📦 Business Type: Exporter | Supplier | OEM Partner
📲 WhatsApp Direct: +91 93599 02383
🔗 Instant Connectivity:




